Abstract
Radioactive iodine (131I) therapy may be used to treat thyroid cancer in end-stage renal disease patients who undergo hemodialysis. Because iodine uses predominantly renal clearance, treatment management in hemodialysis patients may be problematic, and no formal recommendations on hemodialysis currently exist. This work details our experience with treating thyroid cancer with iodine in chronic renal failure patients who require hemodialysis and details the dosimetry results obtained during treatment to ensure that the dose to the bone marrow (BM) was acceptable. Methods: We treated 6 patients in the metabolic radiotherapy unit after thyroid stimulation. Two hemodialysis sessions in the metabolic radiotherapy unit were performed at 42 and 90 h after radiopharmaceutical administration. BM toxicity was estimated with activity measurements from blood samples and with whole-body measurements that were regularly repeated during hospitalization and measured with a γ-counter. The patients underwent thyroid and hematologic monitoring to assess treatment efficacy and therapeutic toxicity in the short, medium, and long term. Results: Whole-body activity was reduced on average by 66.7% (range, 60.1%–71.5%) after the first dialysis session and by 53.3% (range, 30.4%–67.8%) after the second. The mean estimated total absorbed dose to the BM was 0.992 Gy for all patients (range, 0.431–2.323 Gy). We did not observe any significant hematologic toxicity, and the clinical, biologic, and ultrasound test results confirmed the success of ablative treatment for most patients. Conclusion: In hemodialysis patients with thyroid cancer, an 131I activity approximately 30% lower than the nominal dose appears to strike an appropriate balance between absence of BM toxicity and therapeutic efficacy. To avoid overirradiation, we recommend pretherapeutic dosimetry studies for metastatic patients to calculate the amount of activity to be administered. We also recommend dosimetry monitoring during the hemodialysis sessions performed after therapeutic dose administration and under the same conditions.
The management of patients with thyroid cancer usually includes partial or total thyroidectomy with possible lymph node dissection followed by 131I-ablative therapy for incomplete resection or intermediate-risk or high-risk tumor recurrence (1). 131I radiotherapy is therefore recommended for most differentiated thyroid carcinoma patients after complete resection to destroy residues in the thyroid parenchyma (a priori benign), facilitate follow-up care, and treat possible locoregional or distant tumor foci (metastases) (2–4). An extension assessment performed alongside 131I radiotherapy by scintigraphy aims to adapt the management strategy, facilitate biologic monitoring (thyroglobulin) and ultrasound imaging, and improve overall and progression-free survival for these patients (5).
131I is a β- and γ-emitter with a radioactive half-life of 8.1 d. 131I radiation is composed mainly of β-particles with a maximum energy of 606 keV and a mean energy of 192 keV, and γ-rays of 364 keV, allowing for both the treatment of patients and scintigraphic imaging (6,7). After oral administration of 131I, only approximately 20% of the blood iodine is absorbed by the thyroid tissue; the remainder is largely cleared through the urine (up to 75%) (8).
The incidence of cancers, including thyroid cancer (9,10), is relatively higher in patients with chronic renal failure than in patients with healthy kidneys. Although several explanations have been proposed, the pathogenetic mechanism is still not fully understood (11).
Internal radiotherapy using high amounts of 131I activity is difficult in these patients for two reasons (12,13). The first is increased irradiation of the patient (and to close family members and exposed caregivers) arising from the long biologic residence of the isotope due to limited or absent urinary excretion, which risks exceeding the limiting dose to the hematopoietic marrow and causing hematologic toxicities. The second difficulty is the need for a practical facility for dialysis (in a radiation-protected unit or in a hemodialysis department) and adapted management strategies for the radioactive effluents produced during the first few dialysis sessions after iodine therapy.
The nuclear medicine department has dedicated facilities and procedures for monitoring and controlling the risk of exposure to ionizing radiation. However, hemodialysis patients require a controlled dialysis fluid elimination system and highly thorough radioactivity monitoring because the radiopharmaceuticals cannot be eliminated through the renal system. The realization of hemodialysis within the nuclear medicine department allows for optimal radiation protection conditions without the need for human intervention to discard the effluents, which can be directly evacuated to decay tanks. However, currently, very few centers meet the conditions needed to perform hemodialysis sessions from the nuclear medicine department.
Thus far, a limited number of studies have focused on the management of 131I therapy in hemodialysis patients, and currently, no official hemodialysis recommendation exists (14,15).
This work reports our experience with managing 131I treatment for thyroid cancer patients with chronic renal failure who are undergoing hemodialysis and the pretreatment dosimetry results obtained during their treatment.
MATERIALS AND METHODS
Patients
This retrospective study was conducted in the nuclear medicine department of the Roger Salengro Hospital of Lille University Hospital and performed in accordance with the Declaration of Helsinki and national regulations. Since the procedures described were performed as the standard of care, the institutional review board approved this retrospective study, and the requirement to obtain informed consent was waived.
From 2011 to 2017, we included hemodialysis patients with end-stage renal disease and thyroid cancer for whom systemic 131I treatment was recommended after a multidisciplinary team meeting involving specialists in nuclear medicine, endocrinology, and nephrology. The diagnosis of thyroid cancer was confirmed by an anatomopathologic examination of the operative specimen after thyroidectomy. 131I activity prescription was reduced from the nominal dose (i.e., 3,700 MBq) and validated by the multidisciplinary team according to the risk of relapse. When considering a high risk of relapse, the activity ranged from 100% to 60% of the nominal dose (i.e., 3,700–2,200 MBq). When considering a low risk of relapse, the activity was reduced to 50% of the nominal dose (i.e., 1,850 MBq).
Before iodine therapy was systematically scheduled, a consultation took place to examine the patient; confirm the absence of contraindications to the treatment; explain the course of hospitalization, including the dosimetry study; and explain the radiation protection instructions. In women of childbearing age, even though the risk of pregnancy was low, the absence of pregnancy and breastfeeding was verified, and the need for effective contraception for 6–12 mo after treatment was explained.
Hospitalization
Optimal 131I uptake to the thyroid parenchymal tissue (follicular cells) requires prior stimulation with thyroid-stimulating hormone (TSH). This stimulation may be a thyroid hormone withdrawal for at least 4 wk or intramuscular injections of recombinant human TSH (thyrotropin alfa [Thyrogen; Genzyme]).
The standard thyrotropin alfa regimen in patients with preserved renal function is 2 intramuscular injections (or subcutaneous injections if the patient is taking anticoagulants) of 0.9 mg of thyrotropin alfa spaced 24 h apart, with the first injection 2 d before and the second injection 1 d before 131I administration. In hemodialysis patients, a single injection of thyrotropin alfa was performed 48 h before treatment to avoid an excessive TSH level and prolonged period of TSH elevation. The high TSH levels already observed in dialysis patients are associated with side effects such as headache or diarrhea.
The patients were hospitalized for 5 d to allow for dosimetry studies and dialysis in shielded rooms of the nuclear medicine department. Portable hemodialysis equipment was installed in the patient’s room.
On the day of treatment, the patients underwent a dialysis session in the nephrology department before being transferred to the nuclear medicine department. After admission into the metabolic radiotherapy unit, an iodine capsule was orally administered to the patient by the referring physician (the administration was after urination in case of residual diuresis). The day and time of administration and the activity ingested were carefully noted. The capsule was systematically distanced from any factors that could influence the absorption of 131I (particularly gastric protective treatments).
131I whole-body scintigraphy was performed 3 d after treatment with a Symbia T series γ-camera (Siemens Healthcare) to conduct the locoregional extension assessment of the thyroid remnants and cervical ganglionic foci or distant foci (metastases).
When leaving the nuclear medicine department, the patients received radiation protection instructions again in a document that specified the dose rate at 1 m measured on the day of discharge. The main instructions were to avoid prolonged contact with pregnant women and children under 10 y old for the few days after the administration of 131I and to reinforce usual daily hygiene habits.
Before the hemodialysis sessions, regulatory controls of the water circuit were repeatedly performed by the biomedical engineering department to ensure consistency. All our patients had arteriovenous shunts so that they could benefit from regular hemodialysis sessions. Before being hospitalized in the nuclear medicine department, the patients underwent a hemodialysis session of 3–4 h in the hemodialysis department of Lille University Hospital. Subsequently, the patients underwent 2 hemodialysis sessions using a transportable dialysis machine in the nuclear medicine department under the guidance of nurses trained in hemodialysis.
The dialysates were directly eliminated through connections to the decay tanks before being released into the public sewage treatment system (after ∼4–5 mo of decay). The disposable materials used during the hemodialysis sessions were managed as typical radioactive waste of the department. Exposure of the staff to radiation was monitored using passive and operational dosimeters worn at chest level. Our radiation protection protocol for staff members is in accordance with the regulations issued by the French Nuclear Safety Authority. The radiation limit for nonoccupationally exposed personnel (e.g., dialysis nurse) and for the general public is 1 mSv/y, and the exposure limit for nuclear medicine staff is 6 mSv/y.
During hospitalization in the metabolic radiotherapy unit, patients underwent blood tests for thyroglobulin, antithyroglobulin antibodies, and TSH once or twice daily to study their respective kinetics directly after the administration of 131I, particularly in the context of hemodialysis. In parallel, blood counts were performed before and after treatment to verify the absence of hematologic toxicity that may occur after the administration of 131I.
Estimation of the Bone Marrow (BM) Absorbed Dose
According to recommendations, the acceptable absorbed dose to the BM is limited to 2 Gy. The BM dose calculation is complex, but the dose to the blood, which is easier to determine, is an accepted and satisfactory substitute (16). Thus, for the thyroid cancer treatments, given the different radioactive emissions of 131I, the blood (target) is irradiated either by β-particles emitted by the activity of the blood itself (first source) or by penetrating γ-radiation from the activity dispersed through the rest of the body (second source). Therefore, only blood and whole-body measurements should be monitored for radioactivity.
The method used in this study to calculate the absorbed dose to the BM is based on the MIRD formalism (17), which was initially reported in the European Association of Nuclear Medicine Dosimetry Committee series on standard operating procedures for BM dosimetry in the treatment of differentiated thyroid cancer (16,18). This method requires mathematic modeling and a direct measurement of the concentration of radionuclide activity as a function of time in the patient’s body and enables dose calculation using Equation 1: Eq. 1where
Eq. 1where  (Gy) is the mean absorbed dose to the blood;
(Gy) is the mean absorbed dose to the blood; 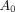 (GBq) is the administered activity;
(GBq) is the administered activity; 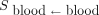 (Gy⋅mL⋅GBq−1⋅h−1) is the S value from the MIRD formalism and is used to estimate blood self-irradiation (only β− contributions were considered, and the value from a previous publication (19) was used:
(Gy⋅mL⋅GBq−1⋅h−1) is the S value from the MIRD formalism and is used to estimate blood self-irradiation (only β− contributions were considered, and the value from a previous publication (19) was used: 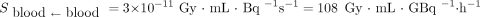 );
);  (h⋅mL−1) is the residence time per milliliter of blood for the activity concentration in the blood (activity normalized to the administered activity) (Eq. 4);
(h⋅mL−1) is the residence time per milliliter of blood for the activity concentration in the blood (activity normalized to the administered activity) (Eq. 4); 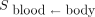 (Gy⋅GBq−1⋅h−1) is the S value used to estimate the contribution of the whole-body activity concentration to the absorbed dose to the blood (only γ-photon contributions are considered); and
(Gy⋅GBq−1⋅h−1) is the S value used to estimate the contribution of the whole-body activity concentration to the absorbed dose to the blood (only γ-photon contributions are considered); and 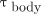 (h) is the residence time in the whole body (Eq. 4).
(h) is the residence time in the whole body (Eq. 4).
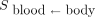 is given by Equation 2:
is given by Equation 2: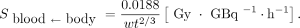 Eq. 2
Eq. 2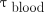 and
and  were estimated from
were estimated from 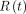 (the fraction of the administered activity
(the fraction of the administered activity  as a function of time t) by means of, respectively, blood sample collection and whole-body acquisition, as shown in Equation 3:
as a function of time t) by means of, respectively, blood sample collection and whole-body acquisition, as shown in Equation 3: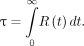 Eq. 3R(t) is measured both for blood samples (
Eq. 3R(t) is measured both for blood samples (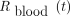 ) and for whole-body acquisitions (
) and for whole-body acquisitions (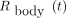 ) such that
) such that  and
and 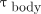 can be computed before estimating the dose from Equation 1.
can be computed before estimating the dose from Equation 1.
As recommended previously (16), to determine 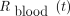 (Eq. 4), we repeatedly collected 2-mL samples of whole-blood during hospitalization in the nuclear medicine department. Whole-body measurements were also taken by a γ-counter (Wallac Wizard 1480; WallacOy).
(Eq. 4), we repeatedly collected 2-mL samples of whole-blood during hospitalization in the nuclear medicine department. Whole-body measurements were also taken by a γ-counter (Wallac Wizard 1480; WallacOy).
To determine the activity concentration of each sample, 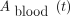 , the number of counts per minute given by the γ-counter, was first corrected from the background noise. Then, the activity per tube was determined from the known calibration factor of the well counter used. Finally, each value obtained was corrected for the physical decay of 131I to account for the time between sampling and analysis and was used to compute
, the number of counts per minute given by the γ-counter, was first corrected from the background noise. Then, the activity per tube was determined from the known calibration factor of the well counter used. Finally, each value obtained was corrected for the physical decay of 131I to account for the time between sampling and analysis and was used to compute 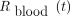 with respect to the activity administered, as shown in Equation 4:
with respect to the activity administered, as shown in Equation 4: Eq. 4The whole-body fraction of the administered activity,
Eq. 4The whole-body fraction of the administered activity, 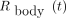 , was determined as shown in Equation 5 using a γ-counter 2 m from the thyroid area. The patient sat on a stool so that the whole body was included in the solid angle of the γ-counter (Atomlab 930; Biodex Medical Systems, Inc.). Anterior and posterior acquisitions of 1 min each were considered for counting γ-photons in the range of 309–419 keV:
, was determined as shown in Equation 5 using a γ-counter 2 m from the thyroid area. The patient sat on a stool so that the whole body was included in the solid angle of the γ-counter (Atomlab 930; Biodex Medical Systems, Inc.). Anterior and posterior acquisitions of 1 min each were considered for counting γ-photons in the range of 309–419 keV: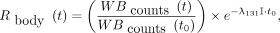 Eq. 5where
Eq. 5where  is the total number of counts at the first acquisition,
is the total number of counts at the first acquisition, 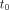 is the time of the first acquisition, and t = 0 is the time of activity administration.
is the time of the first acquisition, and t = 0 is the time of activity administration.
In practice, different procedures describing the chronology, patient position, measurements, data collection, sample analysis, and data recording were implemented in the nuclear medicine department. The different tasks were shared among nurses, medical physicists, and radiopharmacists. Our chronology included blood sampling and body measurements before and 2 h after iodine administration. Subsequently, blood and whole-body measurements were repeated at 6, 24, 40, 50, 72, 88, and 96 h after administration of the 131I capsule.
Table 1 summarizes the chronology of the different radioactive measurement tasks performed during the hospitalization period.
Chronology of Different Tasks Performed
Posttreatment Follow-up
Although thyroid cancer recurrence remains rare, most relapses occur within 5 y of diagnosis, but they may be delayed; thus, an extended follow-up period is required (20).
Surveillance is based primarily on cervical ultrasound and measurement of serum thyroglobulin level (which normally becomes undetectable after 131I ablative therapy in the absence of residual disease or recurrence) and antithyroglobulin antibody level.
In our population, the long-term therapeutic efficacy was evaluated clinically by regular endocrinologic consultations, morphologically by cervical ultrasound in the months after the administration of iodine (on average, 9 mo after), and biologically with regular monitoring of thyroglobulin, antithyroglobulin antibodies, and TSH levels. Chest CT scans were performed to morphologically monitor the presence of thyroid lung metastases. We did not perform systematic scintigraphy assessments in our population.
In parallel with monitoring therapeutic effectiveness, a surveillance of the possible toxicities, particularly hematologic toxicities, was conducted. Specifically, to reflect these possible hematologic toxicities at both the acute and the chronic levels, we recorded the short-term (∼2–6 mo after radioiodine therapy), medium-term (∼1 y), and long-term (at the most recent follow-up) data for each patient.
RESULTS
Patients
We included 6 hemodialysis patients with thyroid cancer who underwent 131I ablation. One of these patients received a second cycle of 131I treatment (6 mo after the first treatment) due to metastatic bone disease. One patient was stage N+ during the initial extension assessment, and 2 were stage M+. The main characteristics of the patients are reported in Table 2.
Main Characteristics of Patients
Course of Therapeutic Management
The pretherapeutic thyroid stimulation for 5 patients consisted of an intramuscular injection of 0.9 mg of recombinant human thyrotropin 48 h before capsule administration, and patient 2 had a thyroid hormone withdrawal 1 mo before treatment (the administered activity and thyroid hormone withdrawal reported for patient 2 in Table 3 was validated by the multidisciplinary team of the Gustave Roussy Institute).
131I Activity Administered, Stimulation Modality, and Characteristics of Dialysis Sessions Performed in Protected Area
The evolution kinetics of blood TSH in patients during their hospitalization is shown Figure 1, and the respective TSH values are shown in Table 4.
Kinetics of blood serum TSH levels during hospitalization).
Blood TSH Values During Hospitalization (in μIU/mL)
Patient 3 received 2 injections of thyrotropin alfa despite our recommendations, and patient 5, who had the highest TSH levels, did not fully adhere to her treatment and had a TSH baseline close to 400 μIU/mL.
All patients were hospitalized for 5 d in our department and received an average 131I activity dose of 2,603 MBq (median, 2,274 MBq; range, 1,842–3,747 MBq). The 2 hemodialysis sessions performed in our department were spaced 2 d apart; most sessions lasted approximately 4 h, with an average total purified blood volume of 75 L.
The main methods of 131I administration and the main characteristics of the hemodialysis sessions are reported in Table 3 (except for patient 6, who had missing dialysis data).
Each patient underwent diagnostic scintigraphy 3 d after 131I administration. Only 1 patient (patient 2 during his first treatment) had distant disease in the form of a bone metastasis on the rib cage (not confirmed by CT or MRI). The scintigraphy images of the anterior and posterior views and SPECT/CT scan of this patient are shown Figure 2.
Scintigraphy and 131I SPECT/CT images of patient 2 after first iodine administration (A) and corresponding images at day 3 after second administration (B), which show disappearance of metastatic focus on left side of 11th rib.
The scans of the other 5 patients did not reveal any secondary focus. Regarding the exposure of staff members, the mean effective dose recorded with the operational dosimeter was 47.7 μSv (range, 30.0–74.0 μSv) during the first session and 11.0 μSv (range, 1.0–22.0 μSv) during the second session.
Evaluation of Therapeutic Efficacy
The results of clinical, biologic (thyroglobulin, antithyroglobulin antibodies, TSH), and ultrasound (of the thyroid) follow-up tests confirmed the success of 131I ablation treatment for 3 of 4 patients classified as M0.
Patient 1 died of an intercurrent disease (nonrevascularizable ischemic heart disease) shortly after 131I treatment and was therefore unable to benefit from a long clinical–biologic follow-up or thyroid ultrasound.
Twelve months after treatment, the thyrotropin alfa test results of patient 4 showed a stimulated thyroglobulin value of less than 1 ng/mL. He underwent kidney transplantation in March 2015 and died 18 mo later from infectious complications under immunosuppression.
In 2018, 1 y after 131I treatment, the thyrotropin alfa test of patient 3 confirmed biologic remission with a stimulated thyroglobulin value of less than 1 ng/mL and the absence of antithyroglobulin antibodies. The patient struggled to comply with hormonal treatment and had VACTERL syndrome (vertebral defects, anal atresia, cardiac defects, tracheoesophageal fistula, renal anomalies, and limb abnormalities), which causes malabsorption and high thyroid hormone requirements (levothyroxine at a dose of 7.5 μg/kg instead of 1.9 μg/kg), as well as difficulties in balancing TSH levels.
Patient 2, who had secondary bone lesions, benefited from a second 131I administration. When whole-body thyroid scintigraphy was performed on day 3 after the second administration, the bone lesions were no longer visible (Fig. 2).
Table 5 shows the results of the thyroid examinations performed before treatment and during the postradioiodine therapy follow-up.
Thyroid Assessments Before and After Treatment
Assessment of Therapeutic Toxicity
None of the patients had any clinically significant side effects (including nausea, vomiting, dry syndrome, or salivary stones). To best reflect acute and chronic toxicities on hematopoiesis from a biologic point of view, we performed blood tests shortly after 131I administration (2–6 mo later), 1 y later, and at the most recent follow-up visit. Over these time points, we did not find any significant decreases in platelet or leukocyte counts or in hemoglobin levels, although this result remains difficult to interpret given the low values of hemoglobin in hemodialysis patients. The overview of blood counts before and after the patients received iodine therapy is reported in Table 6.
Comparison of Hematopoietic Function Before and After Administration
The kinetics of whole-body 131I activity measured with a γ-counter over time are shown in Figure 3. Table 7 presents the values of whole-body activity measured with a γ-counter before and after the 2 hemodialysis sessions in the protected area. The whole-body measurements of patient 6 were obtained with a γ-camera instead of a γ-counter (not available at the time of measurement).
Normalized R(t) activity of whole body as measured by γ-counter as function of time.
Normalized Whole-Body Activity R(t) Measured with γ-Counter Before and After Dialysis and Total Absorbed Dose to Blood Estimated from Measurement
Therefore, the whole-body activity was reduced on average by 66.7% (range, 60.1%–71.5%) after the first dialysis and by 53.3% (range, 30.4%–67.8%) after the second dialysis.
Figure 4 shows the kinetics of blood activity measured via the various blood samples taken during hospitalization in the protected area.
Normalized Rblood(t) for blood activity as function of time (in hours).
The BM dose estimated by the method described in the European Association of Nuclear Medicine standard operating procedures (16,18) using the collected data (Figs. 3 and 4) averaged 0.992 Gy for all patients. The details of the estimated BM total absorbed doses of each patient are presented in Table 7.
The patients were discharged from the hospital with an estimated mean dose rate of 15.0 μSv/h at 1 m (range, 7.0−20.0 μSv/h) and 5.8 μSv/h at 2 m (range, 2−10 μSv/h).
DISCUSSION
The treatment of thyroid cancer with 131I is intended to provide a sufficient dose to the residual thyroid tissue or secondary lesions while avoiding acute or subacute hematologic toxicity in patients with adequate renal function. Although patients 3 and 4 had a low recurrence risk, 131I treatment was achieved because of a kidney grafting project requiring minimization of the risk of relapse to as low as possible.
Finally, only 1 patient (patient 6) had an estimated BM total absorbed dose greater than 2 Gy. Before treatment, the patient benefited from a dosimetry evaluation (use of a low activity to estimate the residence time of 131I in the body with the same methodology). This pretherapy evaluation was achieved with 2-way dialysis, and the BM dose was estimated to be 1.18 Gy for a prescription activity of 3,700 MBq. Unfortunately, during the second stage of management for this patient, which was therapy, the first dialysis session was performed with 1 channel instead of the 2 channels used for the pretherapy evaluation, resulting in a decrease in the flow rate and, thus, in the total purified blood volume. However, this patient did not exhibit any biologic complications during follow-up.
In patients with end-stage renal disease, the spontaneous excretion of urine is very limited or totally absent, and the effective half-life of 131I is more than 4 times higher in these patients than in patients with normal renal function (15). It is imperative that the use of ablative doses in these patients be paired with extrarenal purification to limit the dose to the hematopoietic marrow. This strategy is also recommended by the European Association of Nuclear Medicine Dosimetry Committee. However, that committee does not yet specify the standard dose to be administered, the radioactivity measurement methods to be used, or the methods to be applied for hemodialysis sessions (6).
These results show that hemodialysis sessions for patients treated with 131I are safely and effectively achievable in a shielded area within the nuclear medicine department. Clearly, a good facility infrastructure is mandatory to ensure the quality of the water and of the effluent removal system in the decay tanks. In addition, good communication with the hemodialysis department and the dialysis nursing staff is needed to coordinate the use of mobile dialysis devices.
Unfortunately, the size of our population remains insufficient to draw conclusions, but a single dose of thyrotropin alfa allowed us to obtain satisfactory results in terms of thyrotropic stimulation while offering tangible comfort to the patients by avoiding the issue of hypometabolism related to thyroid hormone withdrawal. The residence time of 131I in dialysis patients does not seem to be related solely to the thyrotropic stimulation modality but is also related to the rhythm and efficiency of the dialysis. Our small number of patients, including only 1 thyroid hormone withdrawal patient with a single distant bone lesion, does not allow us to compare the differences in irradiation according to the type of stimulation or the metastatic and nonmetastatic characteristics of the disease.
There are 2 general approaches to determining the appropriate 131I dose to treat thyroid cancer. Individual dosimetry studies were performed according to previous studies (8,21,22) such as that by Hänscheid et al. (23), who incorporated an analysis based on the MIRD parameter, whereas others used empiric dose methods (12,24–27). Among those, some authors suggested reducing the amount of activity to avoid excessive radiation exposure (12,26), whereas others proposed increasing the amount of activity because of the rapid clearance of iodine with dialysis (25,27). A third point of view suggests that the dose should be the same as that in the population with normal renal function (21,22).
Because patient 6 received administered activity that was similar to the nominal activity and had an estimated BM total absorbed dose that was slightly greater than 2 Gy because of a dialysis exception, we continue to empirically reduce the administered activity of 131I. This activity reduction has allowed nonmetastatic patients to meet the remission criteria while remaining safe in the event of slight deviations from the dialysis protocol.
For patient 2, with bone metastasis, a fixed activity of 3,100 MBq also allowed for the elimination of the bone lesion without causing overirradiation.
Thus, we recommend a 30% reduction in activity from the nominal dose for ablative or adjuvant treatments in hemodialysis patients. However, for metastatic patients, the realization of a pretherapeutic dosimetry study is expected to refine the amount of activity to be administered. Notably, the dialysis sessions must be performed under the exact same conditions as those during the dosimetry studies achieved before or during the 131I radiotherapy to avoid variations such as those in the case of patient 6. Indeed, for this patient, a different dialysis approach led to an erroneous final dosimetry computation, but no hematologic toxicity has been observed in the medium and long term.
The dialysis sessions limit whole-body overirradiation for the patient because of the low level of binding between proteins and iodine, which can easily be removed through hemodialysis membranes. However, the timing of these sessions needs to be correct to allow for dialysis sufficient to prevent the accumulation of excessive irradiation while avoiding too-early dialysis that might eliminate too much iodine, which would reduce the efficacy (8).
Howard et al. (24) showed that for a patient on hemodialysis, the thyroid uptake is 6% at 24 h and 10% at 48 h. Therefore, waiting 42 h after iodine administration for the first dialysis session seemed appropriate.
At patient discharge, the average dose rate at a distance of 1 m was 15.0 μSv/h. This figure is consistent with that found by other authors, such as Murcutt et al. (28), who observed an average dose of less than 20, at 1 m when the patient was discharged from the hospital.
The patient’s whole-body activity was measured before and after each hemodialysis session to assess the clearance of 131I. The mean fraction of 131I cleared in our population was 66.7% (range, 60.1%–71.5%) after the first session. This percentage is consistent with the data reported by other authors, who showed a clearance ranging from 50% to 72% (8,25).
The staff member with the most exposure was the dialysis nurse at the first session. The level of exposure was acceptable for a typically unexposed worker.
In contrast to patients with normal renal function, the 131I elimination curve for renal failure patients on dialysis was not exponential. For this reason, the method we used to calculate the BM dose differs from that used for patients with normal renal function and allows for iodine clearance over a longer period requiring more blood samples and body measurements, especially before and after each dialysis session. None of the patients showed any hematologic toxicity during short-, medium-, or long-term follow-up periods.
Given the rarity of thyroid cancer in hemodialysis patients with renal insufficiency and the limited data in the current literature, we believe that reaching a formal consensus on the ideal management of these patients will be difficult. This situation will likely persist until a significant amount of dosimetry data are accumulated and new studies are undertaken. Finally, the fundamental intention of this study was not to suggest a new therapeutic model or to propose a dose optimization method for these patients but to aggregate data from several actual cases by proposing an effective method to track dose.
CONCLUSION
Our treatment protocol with 131I in patients with thyroid cancer undergoing hemodialysis was safe and efficient. The treatment of thyroid cancer in patients with renal insufficiency who are undergoing hemodialysis requires a multidisciplinary approach involving endocrinologists, physicians, radiopharmacists, nurses, and medical physicists, as well as nephrologists and a dialysis team. This method for determining the absorbed dose might also be useful before therapy to determine the level of 131I activity that should be administered, particularly for metastatic patients. However, this approach for determining the absorbed dose in the BM should be systematically applied for all patients both for monitoring and for ensuring that the absorbed dose remains lower than the 2-Gy threshold for the BM.
DISCLOSURE
No potential conflict of interest relevant to this article was reported.
KEY POINTS
QUESTION: Is the management of patients who have end-stage renal disease and are undergoing hemodialysis alongside their radioiodine therapy achievable?
PERTINENT FINDINGS: From 2011 to 2018, 6 patients were treated in the metabolic radiotherapy unit after thyroid stimulation and with a dedicated protocol to assess the absorbed dose to the BM. The administered activity was reduced by approximately 30%.
IMPLICATIONS FOR PATIENT CARE: The success of ablative treatment for most patients with no significant hematologic toxicity confirmed the possibility of delivering 131I treatment alongside hemodialysis.
Footnotes
↵* Contributed equally to this work.
Published online Jan. 10, 2020.
- © 2020 by the Society of Nuclear Medicine and Molecular Imaging.
REFERENCES
- Received for publication June 5, 2019.
- Accepted for publication December 19, 2019.











