Abstract
Objective
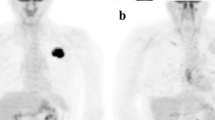
The aim of this study was to clarify the most appropriate follow-up initiation time point for positron emission tomography (PET)/computed tomography (CT) following radio frequency ablation (RFA) of lung tumors, and the cutoff values of maximum standard uptake value (SUVmax) to evaluate local tumor progression.
Methods
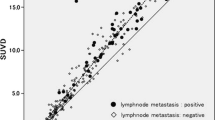
We enrolled 15 patients (8 men, median age 62 years) with 60 tumors, who were treated with RFA of lung tumors and underwent fluorodeoxyglucose (FDG)-PET/CT following RFA. Local tumor progression was assessed by periodic chest CT images prior to and following intravenous administration of a contrast medium. The SUVmax of three periods, namely, 0–3 months, 3–6 months, and 6–9 months after RFA, was evaluated. The appropriate time point for follow-up initiation and the cutoff value of SUVmax were determined using receiver-operating characteristic (ROC) analysis.
Results
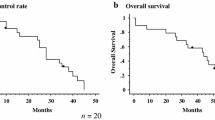
The median follow-up period was 357 days. Of 60 tumors, 10 showed local progression. The area under the ROC curve (Az) for the 6–9 months (P = 0.044) was the largest and almost equal to that of the 3–6 months (P = 0.024). Az for the 0–3 months was the smallest and statistically insignificant (P = 0.705). The cutoff value of 1.5 of SUVmax at 3–9 months after RFA showed 77.8% sensitivity and 85.7–90.5% specificity.
Conclusions
The appropriate follow-up initiation time point is at least 3 months following RFA. Thus, SUVmax is a useful and reliable predictive indicator.
Similar content being viewed by others
References
Mulier S, Ni Y, Jamart J, Ruers T, Marchal G, Michel L. Local recurrence after hepatic radiofrequency coagulation: multivariate meta-analysis and review of contributing factors. Ann Surg 2005;242:158–171.
Komorizono Y, Oketani M, Sako K, Yamasaki N, Shibatou T, Maeda M, et al. Risk factors for local recurrence of small hepatocellular carcinoma tumors after a single session, single application of percutaneous radiofrequency ablation. Cancer 2003;97:1253–1262.
Farrell MA, Charboneau WJ, DiMarco DS, Chow GK, Zincke H, Callstrom MR, et al. Imaging-guided radiofrequency ablation of solid renal tumors. Am J Roentgenol 2003;180:1509–1513.
Burak WE Jr, Agnese DM, Povoski SP, Yanssens TL, Bloom KJ, Wakely PE, et al. Radiofrequency ablation of invasive breast carcinoma followed by delayed surgical excision. Cancer 2003;98:1369–1376.
Hiraki T, Yasui K, Mimura H, Gobara H, Mukai T, Hase S, et al. Radiofrequency ablation of metastatic mediastinal lymph nodes during cooling and temperature monitoring of the tracheal mucosa to prevent thermal tracheal damage: initial experience. Radiology 2005;237:1068–1074.
Yasui K, Kanazawa S, Sano Y, Fujiwara T, Kagawa S, Mimura H, et al. Thoracic tumors treated with CT-guided radiofrequency ablation: initial experience. Radiology 2004;231:850–857.
Lee JM, Jin GY, Goldberg SN, Lee YC, Chung GH, Han YM, et al. Percutaneous radiofrequency ablation for inoperable non-small cell lung cancer and metastases: preliminary report. Radiology 2004;230:125–134.
Steinke K, King J, Glenn DW, Morris DL. Percutaneous radiofrequency ablation of lung tumors with expandable needle electrodes: tips from preliminary experience. Am J Roentgenol 2004;183:605–611.
Hiraki T, Tajiri N, Mimura H, Yasui K, Gobara H, Mukai T, et al. Pneumothorax, pleural effusion, and chest tube placement after radiofrequency ablation of lung tumors: incidence and risk factors. Radiology 2006;241:275–283.
Goldberg SN, Grassi CJ, Cardella JF, Charboneau JW, Dodd GD III, Dupuy DE, et al. Image-guided tumor ablation: standardization of terminology and reporting criteria. Radiology 2005;235:728–739.
de Baère T, Palussière J, Aupèrin A, Hakime A, Abdel-Rehim M, Kind M, et al. Midterm local efficacy and survival after radiofrequency ablation of lung tumors with minimum follow-up of 1 year: prospective evaluation. Radiology 2006;240:587–596.
Steinke K, King J, Glenn D, Morris DL. Radiologic appearance and complications of percutaneous computed tomography-guided radiofrequency-ablated pulmonary metastases from colorectal carcinoma. J Comput Assist Tomogr 2003;27:750–757.
Oyama Y, Nakamura K, Matsuoka T, Toyoshima M, Yamamoto A, Okuma T, et al. Radiofrequency ablated lesion in the normal porcine lung: long-term follow-up with MRI and pathology. Cardiovasc Intervent Radiol 2005;28:346–353.
Yi CA, Lee KS, Kim BT, Choi JY, Kwon OJ, Kim H, et al. Tissue characterization of solitary pulmonary nodule: comparative study between helical dynamic CT and integrated PET/CT. J Nucl Med 2006;47:443–450.
Mac Manus MP, Hicks RJ, Matthews JP, McKenzie A, Rischin D, Salminen EK, et al. Positron emission tomography is superior to computed tomography scanning for response-assessment after radical radiotherapy or chemoradiotherapy in patients with non-small-cell lung cancer. J Clin Oncol 2003;21:1285–1292.
Weber WA, Petersen V, Schmidt B, Tyndale-Hines L, Link T, Peschel C, et al. Positron emission tomography in non-small-cell lung cancer: prediction of response to chemotherapy by quantitative assessment of glucose use. J Clin Oncol 2003;21:2651–2657.
Okuma T, Okamura T, Matsuoka T, Yamamoto A, Oyama Y, Toyoshima M, et al. Fluorine-18-fluorodeoxyglucose positron emission tomography for assessment of patients with unresectable recurrent or metastatic lung cancers after CT-guided radiofrequency ablation: preliminary results. Ann Nucl Med 2006;20:115–121.
Okuma T, Matsuoka T, Okamura T, Wada Y, Yamamoto A, Oyama Y, et al. 18F-FDG small-animal PET for monitoring the therapeutic effect of CT-guided radiofrequency ablation on implanted VX2 lung tumors in rabbits. J Nucl Med 2006;47:1351–1358.
Yamamoto A, Nakamura K, Matsuoka T, Toyoshima M, Okuma T, Oyama Y, et al. Radiofrequency ablation in a porcine lung model: correlation between CT and histopathologic findings. Am J Roentgenol 2005;185:1299–1306.
Yoshioka T, Yamaguchi K, Kubota K, Saginoya T, Yamazaki T, Ido T, et al. Evaluation of 18F-FDG PET in patients with advanced, metastatic, or recurrent gastric cancer. J Nucl Med 2003;44:690–699.
Bunyaviroch T, Coleman RE. PET evaluation of lung cancer. J Nucl Med 2006;47:451–469.
Patz EF Jr, Lowe VJ, Hoffman JM, Paine SS, Burrowes P, Coleman RE, et al. Focal pulmonary abnormalities: evaluation with F-18 fluorodeoxyglucose PET scanning. Radiology 1993;188:487–490.
Duhaylongsod FG, Lowe VJ, Patz EF Jr, Vaughn AL, Coleman RE, Wolfe WG. Detection of primary and recurrent lung cancer by means of F-18 fluorodeoxyglucose positron emission tomography (FDG PET). J Thorac Cardiovasc Surg 1995;110:130–139.
Hashimoto Y, Tsujikawa T, Kondo C, Maki M, Momose M, Nagai A, et al. Accuracy of PET for diagnosis of solid pulmonary lesions with 18F-FDG uptake below the standardized uptake value of 2.5. J Nucl Med 2006;47:426–431.
Rohren EM, Turkington TG, Coleman RE. Clinical applications of PET in oncology. Radiology 2004;231:305–332.
Keidar Z, Haim N, Guralnik L, Wollner M, Bar-Shalom R, Ben-Nun A, et al. PET/CT using 18F-FDG in suspected lung cancer recurrence: diagnostic value and impact on patient management. J Nucl Med 2004;45:1640–1646.
Bastarrika G, Garcia-Velloso MJ, Lozano MD, Montes U, Torre W, Spiteri N, et al. Early lung cancer detection using spiral computed tomography and positron emission tomography. Am J Respir Crit Care Med 2005;171:1378–1383.
Brown RS, Leung JY, Kison PV, Zasadny KR, Flint A, Wahl RL. Glucose transporters and FDG uptake in untreated primary human non-small cell lung cancer. J Nucl Med 1999;40:556–565.
Zhuang H, Pourdehnad M, Lambright ES, Yamamoto AJ, Lanuti M, Li P, et al. Dual time point 18F-FDG PET imaging for differentiating malignant from inflammatory processes. J Nucl Med 2001;42:1412–1417.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Higaki, F., Okumura, Y., Sato, S. et al. Preliminary retrospective investigation of FDG-PET/CT timing in follow-up of ablated lung tumor. Ann Nucl Med 22, 157–163 (2008). https://doi.org/10.1007/s12149-007-0113-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-007-0113-0