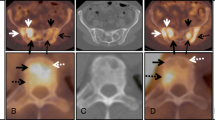
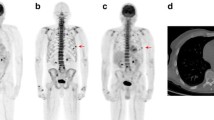
Abstract
Objective
The aim of this prospective study is to evaluate the combined use of fluorine-18 fluorodeoxyglucose (18 F-FDG) and fluorine-18 sodium fluoride (18 F-NaF) PET/CT in the skeletal assessment of patients with multiple myeloma (MM) and to compare the efficacy of these two PET tracers regarding detection of myeloma-indicative osseous lesions.
Patients and methods
The study includes 60 patients with multiple myeloma (MM) diagnosed according to standard criteria. All patients underwent dynamic (dPET/CT) scanning of the pelvis as well as whole body PET/CT studies with both tracers. The interval between the two exams was one day. Sites of focal increased 18 F-FDG uptake were considered as highly suspicious of myelomatous involvement. The lesions detected on the 18 F-NaF PET/CT scans were then correlated with those detected on 18 F-FDG PET/CT, which served as a reference. Moreover, the 18 F-FDG PET/CT results were also correlated with the low-dose CT findings. The evaluation of dPET/CT studies was based on qualitative evaluation, SUV calculation, and quantitative analysis based on a 2-tissue compartment model and a non-compartmental approach.
Results
Whole body 18 F-FDG PET/CT revealed approximately 343 focal lesions while 18 F-NaF PET/CT revealed 135 MM-indicative lesions (39 % correlation). CT demonstrated 150 lesions that correlated with those in 18 F-FDG PET/CT (44 % correlation). Six patients demonstrated a diffuse pattern of disease with 18 F-FDG, while 15 of them had a mixed (diffuse and focal) pattern of skeletal 18 F-FDG uptake. A high number of degenerative, traumatic and arthritic disease lesions were detected with 18 F-NaF PET/CT. In three patients with multiple focal 18 F-FDG-uptake, 18 F-NaF PET/CT failed to demonstrate any bone lesion. The dPET/CT scanning of the pelvic area with 18 F-FDG and 18 F-NaF revealed 77 and 24 MM-indicative lesions, respectively. Kinetic analysis of 18 F-FDG revealed the following mean values: SUVaver = 5.1, k1 = 0.37 (1/min), k3 = 0.10 (1/min), VB = 0.06, influx = 0.04 (1/min), FD = 1.28; the respective values for 18 F-NaF were SUVaverage = 10.7, k1 = 0.25 (1/min), k3 = 0.34 (1/min), VB = 0.02, influx = 0.10 (1/min), FD = 1.37. Apart from the correlation between VB of 18 F-FDG and k1 of 18 F-NaF (r = 0.54), no other significant correlation was observed between the two tracers’ kinetic parameters. We found a significant correlation between FD and SUVaverage (r = 0.93), FD and SUVmax (r = 0.80), FD and influx ( r = 0.85), as well as between influx and SUVaverage (r = 0.74) for 18 F-FDG. In 18 F-NaF we observed the most significant correlations between FD and SUVaverage (r = 0.97), FD and SUVmax (r = 0.87), and between influx and k1 (r = 0.72).
Conclusion
The combined use of 18 F-FDG PET/CT and 18 F-NaF PET/CT provides different molecular information regarding the biological processes that take place in a MM osseous lesion. 18 F-FDG PET/CT proved to be a more specific biomarker than 18 F-NaF PET/CT in multiple myeloma skeletal assessment.




Similar content being viewed by others
References
International Myeloma Working Group. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol. 2003;121:749–57.
Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011;364:1046–60.
Walker RC, Brown TL, Jones-Jackson LB, De Blanche L, Bartel T. Imaging of multiple myeloma and related plasma cell dyscrasias. J Nucl Med. 2012;53:1091–101.
Mason NS, Lin EC. Basics of fluorodeoxyglucose radiochemistry and biology. In: Lin EC, Alavi A (eds). PET and PET/CT: a clinical guide. Thieme medical publisher, Inc 2005; p.15-20.
Durie BG, Waxman AD, D’Agnolo A, Williams CM. Whole-body (18)F-FDG PET identifies high-risk myeloma. J Nucl Med. 2002;43:1457–63.
Schirrmeister H, Bommer M, Buck AK, Müller S, Messer P, Bunjes D, et al. Initial results in the assessment of multiple myeloma using 18 F-FDG PET. Eur J Nucl Med Mol Imaging. 2002;29:361–6.
Bredella MA, Steinbach L, Caputo G, Segall G, Hawkins R. Value of FDG PET in the assessment of patients with multiple myeloma. AJR Am J Roentgenol. 2005;184:1199–204.
Fonti R, Larobina M, Del Vecchio S, De Luca S, Fabbricini R, Catalano L, et al. Metabolic tumor volume assessed by 18 F-FDG PET/CT for the prediction of outcome in patients with multiple myeloma. J Nucl Med. 2012;53:1829–35.
Durie BG. The role of anatomic and functional staging in myeloma: description of Durie/Salmon plus staging system. Eur J Cancer. 2006;42:1539–43.
Blau M, Ganatra R, Bender MA. 18 F-Fluoride for bone imaging. Semin Nucl Med. 1972;2:31–7.
Grant FD, Fahey FH, Packard AB, Davis RT, Alavi A, Treves ST. Skeletal PET with 18 F-fluoride: applying new technology to an old tracer. J Nucl Med. 2008;49:68–78.
Segall G, Delbeke D, Stabin MG, Even-Sapir E, Fair J, Sajdak R, et al. SNM. SNM practice guideline for sodium 18 F-fluoride PET/CT bone scans 1.0. J Nucl Med. 2010;51:1813–20.
Schiepers C, Nuyts J, Bormans G, Dequeker J, Bouillon R, Mortelmans L, et al. Fluoride kinetics of the axial skeleton measured in vivo with fluorine-18-fluoride PET. J Nucl Med. 1997;38:1970–6.
Hawkins RA, Choi Y, Huang SC, Hoh CK, Dahlbom M, Schiepers C, et al. Evaluation of the skeletal kinetics of fluorine-18-fluoride ion with PET. J Nucl Med. 1992;33:633–42.
Nishiyama Y, Tateishi U, Shizukuishi K, Shishikura A, Yamazaki E, Shibata H, et al. Role of 18 F-fluoride PET/CT in the assessment of multiple myeloma: initial experience. Ann Nucl Med. 2013;27:78–83.
Sokoloff L, Smith CB. Basic principles underlying radioisotopic methods for assay of biochemical processes in vivo. In: Greitz T, Ingvar DH, Widén L, editors. The metabolism of the human brain studied with positron emission tomography. New York: Raven Press; 1983. p. 123–48.
Dimitrakopoulou-Strauss A, Strauss LG, Burger C, Mikolajczyk K, Lehnert T, Bernd L, et al. On the fractal nature of positron emission tomography (PET) studies. World J Nucl Med. 2003;4:306–13.
Dimitrakopoulou-Strauss A, Pan L, Strauss LG. Quantitative approaches of dynamic FDG-PET and PET/CT studies (dPET/CT) for the evaluation of oncological patients. Cancer Imaging. 2012;12:283–9.
Pan L, Mikolajczyk K, Strauss L, Haberkorn U, Dimitrakopoulou-Srrauss A. Machine learning based parameter imaging and kinetic modeling of PET data. J Nucl Med. 2007;48:158p.
Strauss LG, Conti PS. The applications of PET in clinical oncology. J Nucl Med. 1991;32:623–48.
Burger C, Buck A. Requirements and implementations of a flexible kinetic modeling tool. J Nucl Med. 1997;38:1818–23.
Mikolajczyk K, Szabatin M, Rudnicki P, Grodzki M, Burger C. A Java environment for medical image data analysis: initial application for brain PET quantitation. Med Inform. 1998;23:207–14.
Miyazawa H, Osmont A, Petit-Taboué MC, Tillet I, Travère JM, Young AR, et al. Determination of 18 F-fluoro-2-deoxy-D-glucose rate constants in the anesthetized baboon brain with dynamic positron tomography. J Neurosci Methods. 1993;50:263–72.
Cheng C, Alt V, Dimitrakopoulou-Strauss A, Pan L, Thormann U, Schnettler R, et al. Evaluation of new bone formation in normal and osteoporotic rats with a 3-mm femur defect: functional assessment with dynamic PET-CT (dPET-CT) using a 2-deoxy-2 [(18)F] fluoro-D-glucose (18 F-FDG) and 18 F-fluoride. Mol Imaging Biol. 2013;15:336–44.
Ohtake T, Kosaka N, Watanabe T, Yokoyama I, Moritan T, Masuo M, et al. Noninvasive method to obtain input function for measuring tissue glucose utilization of thoracic and abdominal organs. J Nucl Med. 1991;32:1432–8.
Strauss LG, Klippel S, Pan L, Schönleben K, Haberkorn U, Dimitrakopoulou-Strauss A. Assessment of quantitative FDG PET data in primary colorectal tumours: which parameters are important with respect to tumour detection? Eur J Nucl Med Mol Imaging. 2007;34:868–77.
Bird JM, Owen RG, D’Sa S, Snowden JA, Pratt G, Ashcroft J, et al. Guidelines for the diagnosis and management of multiple myeloma 2011. Br J Haematol. 2011;154:32–75.
van Lammeren-Venema D, Regelink JC, Riphagen II, Zweegman S, Hoekstra OS, Zijlstra JM. 18 F-fluoro-deoxyglucose positron emission tomography in assessment of myeloma-related bone disease: a systematic review. Cancer. 2012;15(118):1971–81.
Czernin J, Satyamurthy N, Schiepers C. Molecular mechanisms of bone 18 F-NaF deposition. J Nucl Med. 2010;51:1826–9.
Dimitrakopoulou-Strauss A, Strauss LG, Heichel T, Wu H. Burger et al. The role of quantitative (18)F-FDG PET studies for the differentiation of malignant and benign bone lesions. J Nucl Med. 2002;43:510–8.
Acknowledgements
This study is part of the Sonderforschungsbereich-Transregio 79 (SFB-TRR 79) and was financially supported by the Deutsche Forschungsgemeinschaft (German Research Foundation, DFG).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sachpekidis, C., Goldschmidt, H., Hose, D. et al. PET/CT studies of multiple myeloma using 18 F-FDG and 18 F-NaF: comparison of distribution patterns and tracers’ pharmacokinetics. Eur J Nucl Med Mol Imaging 41, 1343–1353 (2014). https://doi.org/10.1007/s00259-014-2721-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-014-2721-y